Clinical Research Studies
Hope for Patients
Our research offers hope for current patients and opportunities to make a difference for future patients. We explore every area of cardiovascular care. Ask your doctor about active MHIF studies that match your treatment needs.
Benefits of Research
Be More Active
Take a more active role in your own health care.
Potential to Unlock More Options
You may gain access to investigational treatments and medications.
Make a Contribution
Help find a new and better way to treat people with your condition.
Promise for Providers
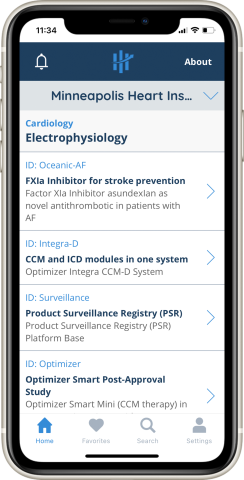
MHIF leads more than 230 studies annually, promising you and your patients access to cutting-edge care. Our High Enroll app makes it even easier to connect with this clinical advantage. By downloading the app and establishing a free account, providers can:
- Follow currently-enrolling MHIF studies
- Learn study details and inclusion/exclusion criteria
- Identify and refer patients
- Receive updates and new study notifications
- Communicate with research coordinators
Our app puts all this information at your fingertips, whether you’re on- or off-line.
As one of the leading nonprofit cardiovascular research and education institutions, MHIF is proud to be the first in Minnesota to bring this innovative app to providers. Join us in raising awareness for life-saving research.
Download High Enroll and establish your account today.
| Download on Google Play |
| Download on Apple Store |
|
Image
|






